
A cosmetic product, which have been tested by the Laboratory of Pharmacy Section, Department of Scientific Services, Ministry of Health was found to be adulterated with undeclared, scheduled poison listed in the Poisons Act 1956.
Glowing Beauty Skincare by Glowing Beauty - Glowing Night Treatment was found to be adulterated with Hydroquinone which can cause adverse effects that are potentially hazardous to the people using them. Unsupervised use of Hydroquinone may cause skin hypersensitivity, skin discoloration resulting in gradual darkening of the affected skin area and an increased risk of skin cancer. The Ministry of Health has not issued any approval for the importation for the sale of the product and/or Cosmetic Notification Acknowledgement Letter for the sale of the affected product.
Following the finding, the product is not allowed to be imported and sold in Brunei Darussalam. Violations to the Act is an offence and the individual may be fined not exceeding $5,000, imprisonment for a term not exceeding two years or both. The public who have purchased or used the products are advised to stop using it immediately. They should also consult a medical practitioner if they feel unwell or experience any undesirable reactions as a result of using the product.
For further information and communication, contact the Compliance and Licensing Section; OR call 239 3301 extension 208; OR e-mail to 'pharmacy.enforcement@moh.gov.bn'; OR contact Talian Darussalam 123.
SOURCE/AUTHOR : RTB NEWS
Venues
Related News

Audience Ceremony with Coordinating Minister for Public Affairs and Minister for Defence, Republic of Singapore

His Royal Highness receives in Audience Coordinating Minister for Public Affairs and Minister for Defence, Republic of Singapore

Congratulatory Messages on Occasion of Malaysia's 68th Anniversary of Independence Day

Congratulatory Messages on the Occasion of the Socialist Republic of Viet Nam's National Day 80th Anniversary

Registration for National Level Musabaqah Al-Qur'an Memorisation and Its Understanding 1447 Hijrah

Course Closing Ceremony, Pengiran Anak Puteri Muta-Wakkilah Hayatul Bolkiah Religious School

Tahlil Ceremony in conjunction with 50th Anniversary of Brunei Arts and Handicrafts Training Centre

Congratulatory Messages on the occasion of Kingdom of Saudi Arabia's 95th National Day

Inauguration Of Establishment of Johor Islamic Religious Council & Multaqa Ulama' Darul Ta'zim

Congratulatory Message on the Occasion of Republic of Korea's National Foundation Day

Doa Kesyukuran Ceremony in conjunction with Her Majesty Duli Raja Isteri's Birthday Celebration

Audience Ceremony with The Secretary of National Defence of the Republic of the Philippines

Khatam Al-Qur'an Ceremony in conjunction with Birthday Celebration of Her Majesty Duli Raja Isteri

Audience Ceremony with Spouse of the Chief of Staff of the Armed Forces of the Philippines

Doa Kesyukuran Ceremony in conjunction with the Birthday Celebration of Her Majesty Duli Raja Isteri

Khatam Al-Qur'an Ceremony in conjunction with the Ruby Jubilee of the Islamic Da'wah Centre

Minister of Foreign Affairs of the Lao People’s Democratic Republic Audience Ceremony

Prizes Presentation, National Preliminary Musabaqah Al-Qur'an Reading Competition for Adults

Khatam Al-Qur'am Ceremony in conjunction with the 50th anniversary of Television Services, RTB

Congratulatory Message on the occasion of the Republic of Turkiye's 102nd Republic Day

Sales Festival in conjunction with the Farmers, Livestock Breeders and Fishermen Day Celebration

Message of Condolences following the Tragic Incident of a Turkish Military Cargo Plane Crash

RB awarded 2026 APEX Four Star Major Airline & 2026 APEX Best in Seat Comfort Award for East Asia

Meeting of the Steering Committee with the Sectoral Committees for the Diamond Jubilee Celebrations

Silver Jubilee Celebration, 25th Anniversary of Yayasan Sultan Haji Hassanal Bolkiah Mosque

His Royal Highness Prince Haji Sufri Bolkiah watches Fencing Event of Islamic Solidarity Games 2025

His Royal Highness Prince Haji Sufri Bolkiah presents the Gold Medals, Islamic Solidarity Games 2025

Certification Application for Religious Teaching, Al-Qur'an, and Lectures 1st Session of 2026

Senior Minister of State, Ministry of Home Affairs Republic of Singapore Audience Ceremony

Ambassador Extraordinary and Plenipotentiary of the People's Republic of China Audience Ceremony

Senior Minister of State, Ministry of Home Affairs Republic of Singapore Audience Ceremony

Ambassador Extraordinary and Plenipotentiary of the People’s Republic of China Audience Ceremony

Launching of Guideline Book on Addressing Harassment at the Workplace in the Public Service

Messages of Condolences following the recent fire at Wang Fuk Court Residential Complex

Congratulatory Messages on the occasion of the National Day of the Kingdom of Thailand

Nine To Four Run in conjunction with 'Ke Temburong Bisai Programme: Borneo Bike Gathering Edition'

Message of Condolences following the Terrorist Attack at Bondi Beach, Sydney, Australia

Their Majesties will attend a Get-Together Ceremony with Citizens and Residents in United Kingdom and Ireland

Get-Together Ceremony with Citizens and Residents of Brunei in United Kingdom and Ireland

Doa Kesyukuran Ceremony in conjunction with the 42nd Anniversary of Kampung Sungai Besar Mosque

Message of Congratulations on the occasion of Republic of the Union of Myanmar's 78th Independence Day

Working Visit by Ministry of Religious Affairs Delegation for the Admission of Sanah 5 Students

Exhibition Match in conjunction with Peak x UNIKLEARN Pickleball Charity Championship 2026

His Majesty will be Taking Rest, Limited Presence at National and Official Ceremonies

Sovereign's Parade Ceremony of the 24th Intake of the Officer Cadet School, Defence Academy, RBAF

Discipline and Self-Resilience are the Core of the Success of 24th Intake of Officer Cadets

Sunat Hajat Prayer, Reading of Surah Yasin and Special Doa, Brunei Darussalam Students Overseas

Official Academic Visit by Muslim Scholars Association of Malaysia at Islamic Da'wah Centre

Commemorative Products Press Conference in conjunction with Diamond Jubilee Wedding Anniversary

Brunei-Cambridge G.C.E. 'O' Level and IGCSE Examination Results for October/November 2025 Session

Agreement Signing between Warisan Shipping Line and DP World Logistics Malaysia Sendirian Berhad

Message of Congratulations on Occasion of the Islamic Republic of Iran's National Day

Tahlil Ceremony and Appointment Letter Presentation, Jubli Perak Sultan Haji Hassanal Bolkiah Mosque

Presentation of Appreciation Certificates and Apointment Letters, Selangan Mosque Takmir Committee

Distribution of His Majesty's Personal Gift of Dates, Ministry of Primary Resources and Tourism

Urgent Treated Water Pump Valve Replacement Works at Bukit Barun Water Treatment Plant Stage 4 & 5

Enforcement of Control of the Capture, Importation of Small-sized and Egg-Bearing Lobsters and Crabs

Congratulatory Message on occasion of Appointment as the New Prime Minister of the People's Republic of Bangladesh

Religious Ceremony Awaiting the Results of the Sighting of the New Moon of Ramadhan 1447 Hijrah

Temporary Half Lane Road Closure at Jalan Raja Isteri Pengiran Anak Saleha and Tungku Highway

Doa Kesyukuran Ceremony in conjunction with His Royal Highness the Crown Prince's 52nd Birthday

Doa Kesyukuran Ceremony in conjunction with the Crown Prince's 52nd Birthday Celebration

Prohibition of Offering Ramadhan Buffet for Premises that have not obtained Halal Certificate

Flag Handover Ceremony to the National Contingent competing at the 6th Asian Beach Games

Brunei's Commitment to Fostering Peace, Promoting Social Harmony & Advancing Sustainable Development

Book Launching Ceremony, Ambassador Extraordinary and Plenipotentiary of the Republic of Turkiye

Audience Ceremony with the Director-General of the Food and Agriculture Organisation of the United Nations, FAO and Delegation

38th Session of Food and Agriculture Organisation Regional Conference Launching Ceremony

Signing of Agreement between Kedai Kampung & Agrodynamics Antarabangsa Brunei Sendirian Berhad

27/04/2026 Opening of the Final of the National Musabaqah Al-Qur'an Reading for Adults 1447 Hijrah/2026

Mass Sunat Hajat Prayer in conjunction with the World Day for Safety and Health at Work 2026

Final of National Level Musabaqah Al-Qur'an Reading for Adults 1447H/2026M, Final Night
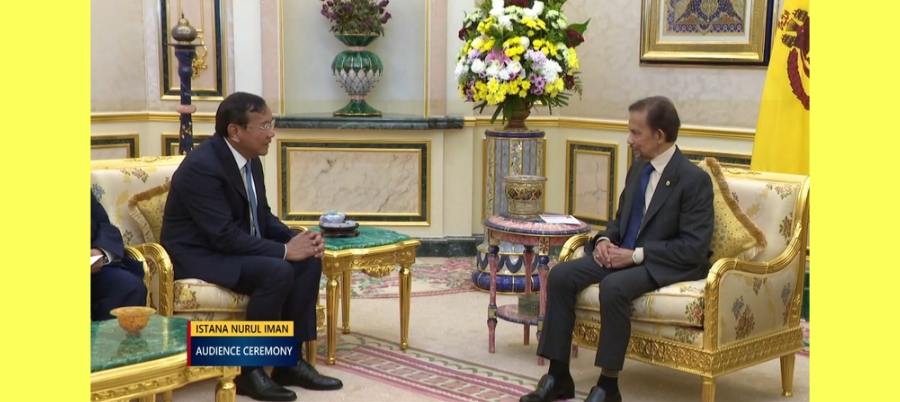
Audience Ceremony with Deputy Prime Minister and Minister of Foreign Affairs and International Cooperation of Cambodia

Conferment of State Honorary Medals in conjunction with His Majesty's 79th Birthday Celebration

Establishment of Tilawah Al-Qur'an Scheme for Children as Foundation for Noble Morals and Personalities

Reading, Deepening Understanding and Expressing Internalisation of Al-Qur'an in Shaping Morals and Behavior

Appointment & Confirmation of Deputy Permanent Secretary (Policy and Management), Ministry of Health

Advance Party of Brunei Darussalam Haj Management Office meeting with Syarikat Holiday Inn Bakkah

Bilateral Meeting With His Excellency Prime Minister of the Democratic Republic of Timor-Leste

Flowmeter and Valve Replacement Works at the Agis-Agis Water Treatment Plant, Belait District

Bilateral Meeting with the President and the Vice President of the People's Republic of China
Brunei Prayer Times
| Suboh | 4:53 am |
| Zohor | 12:17 pm |
| Asar | 3:39 pm |
| Maghrib | 6:27 pm |
| Isyak | 7:36 pm |
Venues
Archive
Lagu HK KDYMM Ke-79: Putri Norizah - Puja Usia (1447H / 2025M) - Versi Terkini













































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































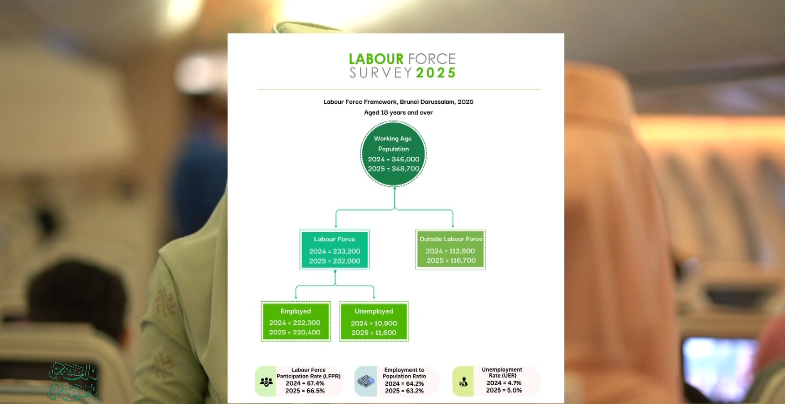























































































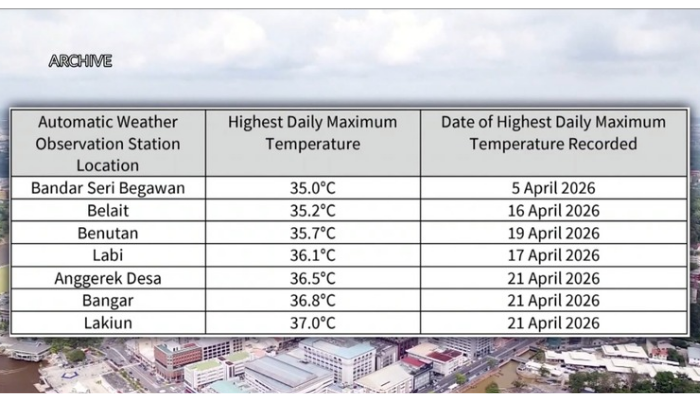































































































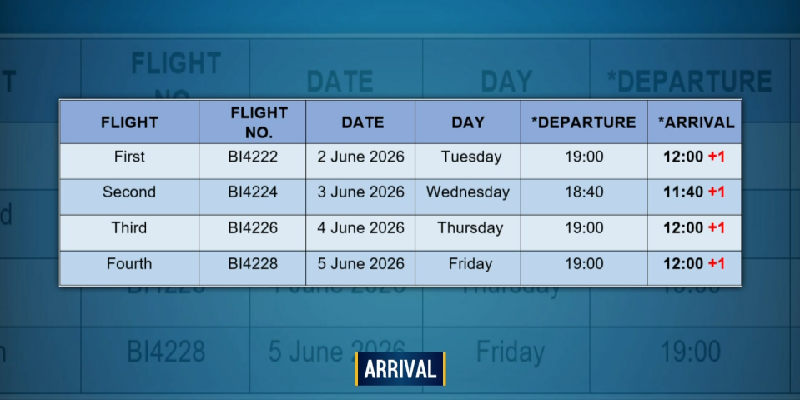
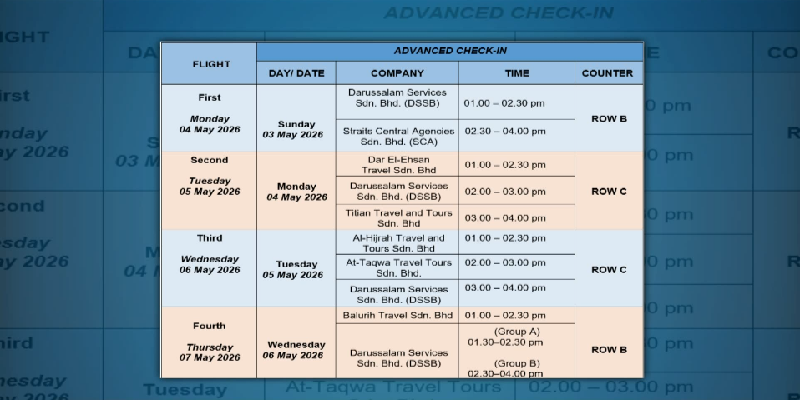

































































































































Comments (0)
No comments yet. Be the first to comment!
Leave a Comment